Inherited causes of clonal haematopoiesis in 97,691 whole genomes
Abstract
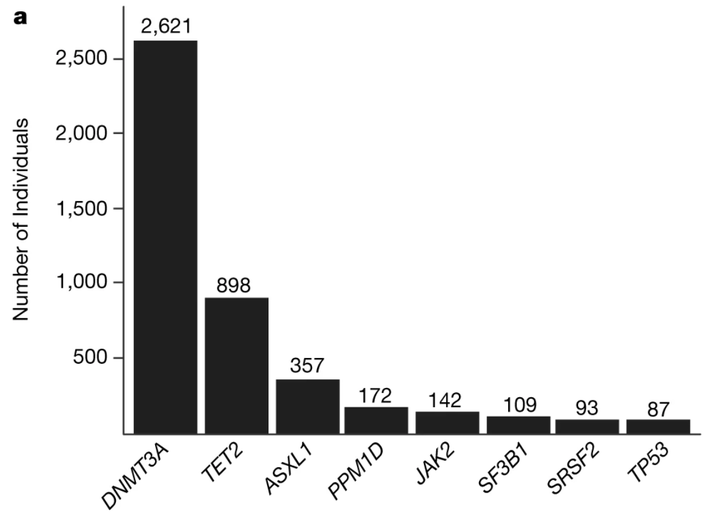
Age is the dominant risk factor for most chronic human diseases, but the mechanisms through which ageing confers this risk are largely unknown. The age-related acquisition of somatic mutations that lead to clonal expansion in regenerating haematopoietic stem cell populations has recently been associated with both haematological cancer and coronary heart disease—this phenomenon is termed clonal haematopoiesis of indeterminate potential (CHIP). Simultaneous analyses of germline and somatic whole-genome sequences provide the opportunity to identify root causes of CHIP. Here we analyse high-coverage whole-genome sequences from 97,691 participants of diverse ancestries in the National Heart, Lung, and Blood Institute Trans-omics for Precision Medicine (TOPMed) programme, and identify 4,229 individuals with CHIP. We identify associations with blood cell, lipid and inflammatory traits that are specific to different CHIP driver genes. Association of a genome-wide set of germline genetic variants enabled the identification of three genetic loci associated with CHIP status, including one locus at TET2 that was specific to individuals of African ancestry. In silico-informed in vitro evaluation of the TET2 germline locus enabled the identification of a causal variant that disrupts a TET2 distal enhancer, resulting in increased self-renewal of haematopoietic stem cells. Overall, we observe that germline genetic variation shapes haematopoietic stem cell function, leading to CHIP through mechanisms that are specific to clonal haematopoiesis as well as shared mechanisms that lead to somatic mutations across tissues.